Question – 1
1. The respiration process of human beings is based on the principle of
-
A. Boyle’s law
-
B. Charles law
-
C. Dalton’s law of partial pressure
-
D. Graham’s law
- E. Both A & B
- Answer:A
- Answer Explanation:During inhalation of gas, pressure inside the lung drops below the air pressure gets decrease and the chest cavity expands. and thus volume gets increased. This shows that pressure and volume are inversely related to each other.
Question – 2
2. Calculate the binding energy of lithium atom (3Li7). The theoretical mass of lithium is 1.172 X 10-26 Kg and the actual mass of lithium is 1.165 X 10-26 Kg
-
A. 6.29 X 10-12 J/atom
-
B. 3.03 X 10-12 J/atom
-
C. 7.10 X 10-13 J/atom
-
D. 6.29 X 10-13 J/atom
- E. 3.03 X 10-13 J/atom
- Answer:A
- Answer Explanation:Binding energy = ??E = ??mc2
??m = (Theoretical mass – actual mass)
= (1.172 X 10-26 – 1.165 X 10-26) = 0.007 X 10-26 Kg
Binding energy = (0.007 X 10-26 Kg) X (2.998 X 108)2 Kg m2 s-2
= 6.29 X 10-12 J/atom
Question – 3
3. What is the mass percentage of carbon in benzene?
-
A. 72 %
-
B. 97 %
-
C. 50 %
-
D. 92 %
- E. 80 %
- Answer:D
- Answer Explanation:Percentage of a certain atom in molecular mass can be calculated by the following formula
Mass Percentge = (Certain atomic mass / Total atomic mass of the molecule)*100
Mass percentage = (6 X 12/78) X 100 = 92 %
Question – 4
4. Identify the name of the mechanism for the following reaction. 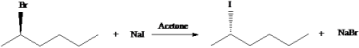
-
A. E2
-
B. E1
-
C. SN1
-
D. SN2
- E. Addition
- Answer:D
- Answer Explanation:The above reaction indicates that it undergoes a Walden inversion. The nucleophile (I–) of the product is behind the plane to the plane of the leaving group (Br–). The reaction takes place in single step mechanism with aprotic solvent. So, it is a SN2 reaction.
Question – 5
5. 3rd qus Repeated
-
A. Repeated
-
B. Repeated
-
C. Repeated
-
D. Repeated
- E. Repeated
- Answer:D
- Answer Explanation:Repeated
Question – 6
6. An azeotropic mixture is also called
-
A. Increased boiling mixture
-
B. Decreased boiling mixture
-
C. Constant boiling mixture
-
D. Either a or b
- E. None of these
- Answer:C
- Answer Explanation:A pure chemical compound boils at a constant temperature and distils over completely at the same temperature without any change in composition. It is also known as constant boiling mixture.
Question – 7
7. The relative intensity of signals in proton NMR is related to
-
A. Chemical shift and magnetic environment of proton
-
B. Different number of protons
-
C. Number of adjacent atoms containing number of protons
-
D. Total number of protons present in the molecule
- E. Coupling constant
- Answer:D
- Answer Explanation:The relative intensity of signals in proton NMR is proportional to total number of protons present in the molecule. Number of signals indicates how many different kinds of protons are present in the molecule. Position of signals indicates the chemical shift and magnetic environment of proton. Splitting of signals indicates the number of adjacent atoms containing different number of protons.
Question – 8
8. Consider aquantity with 44 as its true value. While performing the experiment, the observed value is reported as 43.9. Calculate the relative error.
-
A. 0.22
-
B. 0.025
-
C. 0.00227
-
D. 0.1
- E. 0.5
- Answer:C
- Answer Explanation:Given data: True value = 44, observed value = 43.9
Relative error = Absolute error / True value
Absolute error = True value – observed value = 44 – 43.9 = 0.1
Relative error = 0.1 / 44 = 2.27 * 10-3
Relative error = 0.00227
Question – 9
9. Which of the following is an example for an isolated system?
-
A. A pot of boiling water
-
B. Boiling a soup in an open sauce pan in a stove
-
C. Cooking rice in a pressure cooker
-
D. An open tank of water
- E. Hot water in a thermos flask
- Answer:E
- Answer Explanation:Hot water in a thermos flask is an example for an isolated system where neither energy nor matter can enter or exit. Boiling a soup in an open sauce pan in a stove is an example for open system in which, it can freely exchange its energy and matter with its surroundings. Cooking rice in a pressure cooker is an example for a closed system where it can exchange only energy with its surroundings. A pot of boiling water and an open tank of water is an example for an open system as the matter gets exchanged with its surroundings.
Question – 10
10. Select the incorrect phrase(s) about the significance of salt bridgeI. Connects the solutions of two half-cell reactions.II. It keeps the solution of two half-cells electrically charged.III. Prevents liquid-liquid junction potential.
-
A. I & II
-
B. I & III
-
C. II & III
-
D. II only
- E. III only
- Answer:D
- Answer Explanation:It keeps the solution of two half cells electrically neutral. In anodic half cell, positive ions will accumulate around the anode due to deposition of negative ions by oxidation. To neutralise these positive ions, sufficient number of negative ions are provided by salt bridge. Similarly for cathodic half cell, it is vice-versa.
Question – 11
11. Which element has the greatest tendency to lose electrons?
-
A. F
-
B. Fr
-
C. Be
-
D. S
- E. Ar
- Answer:B
- Answer Explanation:Fr (Francium) has [Rn] 7s1. It is the only element to lose electrons easily because its valency shell has only one electron. By losing one electron it attains the stable noble gas configuration.F (Fluorine) – [He] 2s2 2p5has nearly half filled stable electronic configuration. Be (Beryllium) – [He] 2s2 has fully filled stable s-orbital. S (sulphur) – [Ne] 3s2 3p4 has half filled p-orbital. Ar (Argon) – [Ne] 3s2 3p6 has fully filled p-orbital. So orbitals having half filled or fully filled electrons are more stable and they cannot lose their outermost electrons very easily.
Question – 12
I)Schottky defect is generally observed in ionic compounds with a high coordination number
II) It arises due to displacement of an ion from its lattice point and occupies the interstitial position
III) It’s mainly observed in the compounds, in which the cation and the anion have equal size
-
A. I and II
-
B. I only
-
C. II and III
-
D. II only
- E. I, II and III
- Answer:D
- Answer Explanation:Schottky defect arises due to absence of a set of cation and anion from its lattice site and not by the displacement of cations and anions.
Question – 13
I) The overall rate of the reaction is proportional to the concentration of one of the reactants.
II) If the concentration of the reactant is doubled, the rate is doubled.
III) The unit of rate constant for a first order reaction is sec-1 or time-1.
-
A. I and III only
-
B. I and II only
-
C. I only
-
D. I, II and III
- E. III only
- Answer:D
- Answer Explanation:Rate of a first order reaction, depends only on one of the reactant’s concentrations. When the concentration of the reactant is increased by ‘n’ times, the rate of reaction is also increased by ‘n’ times. The unit of rate constant for the first order reaction is sec-1 or time-1. k1 = rate/ (a-x) = mol.lit-1sec-1 / mol.lit-1 = sec-1
Question – 14
14. The bond order for helium molecule is
-
A. 1
-
B. 2
-
C. 2.5
-
D. 3
- E. 0
- Answer:E
- Answer Explanation:The electronic configuration of helium in the ground state is represented as (??1s)2 and in the excited state, it is represented as (??*1s)2.So, number of electrons in bonding molecular orbital (Nb) is 2 and number of electrons in antibonding molecular orbital (Na) is 2.
Bond order= (Nb-Na)/2 = (2-2)/2 = 0
The bond order for He2 is 0, so the molecule does not exist.
Question – 15
15. Choose the strongest acid from the following.
-
A. HClO4
-
B. HClO3
-
C. HClO2
-
D. HClO
- E. None of the above
- Answer:A
- Answer Explanation:HClO4 is the strongest acid than the rest of the acids. The acidity is due to the presence of the more electronegative oxygen. The acid with ore oxygen is the strongest acid and it has the highest electronegativity. It can easily pull the electrons from the central atom and induces the central atom to draw the electrons from the hydrogenated oxygen. This weakens the O-H bond and H+ ion leaves the molecule easily.