Question – 1
1. Which of the following has higher bond energy?
- A. Methane
- B. Ethene
- C. Acetylene
- D. Oxygen
- E. CO2
- Answer:D
- Answer Explanation:Bond energy is the energy required to break a bond in a molecule. A double bond in a homonuclear diatomic molecule (O=O) has higher bond energy than a single bond (methane) and a triple bond (acetylene)has higher bond length than a double bond between heteronuclear diatomic molecule (CO2).
Question – 2
2. What is the equilibrium constant for the following reaction?
- A. Kc = [Sn][CO2]/[SnO2][CO]2
- B. Kc = [CO2]/[CO]2
- C. Kc = [Sn][CO2]2/[CO]2
- D. Kc = [CO2]2/[CO]2
- E. Kc = [CO2]2
- Answer:D
- Answer Explanation:The equilibrium constant for the above reaction is Kc = [CO2]2/[CO]2. The above reaction is an example for a heterogeneous equilibrium. In heterogeneous equilibrium, the concentrations of the solid and liquid are constant. Therefore, the equilibrium constant doesn’t contain the concentration of the solid and liquid.
Question – 3
3. Find out an unsaturated acid from the following.
- A. 2-Hydroxy propionic acid
- B. 3-Phenyl acrylic acid
- C. Succinic acid
- D. Tartaric acid
- E. Adipic acid
- Answer:B
- Answer Explanation:An unsaturated acid is one which contains a double bond in the molecule. Here, the 3-Phenyl acrylic acid is an unsaturated acid, because it contains a double bond. The rest of the acids are saturated acids, which don’t have a double bond. The structure of 3-Phenyl acrylic acid is as follows,
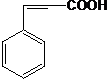
Question – 4
4. The total number of lone pairs of electrons available in BeCl2 and NH3 is
- A. 0, 1 respectively
- B. 2, 0 respectively
- C. 1, 1 respectively
- D. 1, 2 respectively
- E. 2, 2 respectively
- Answer:A
- Answer Explanation:Beryllium chloride has no lone pair of electrons, because beryllium has two valence electrons and they are shared with two chlorine atoms. Ammonia has a lone pair of electrons, because it has five valence electrons in which three electrons are shared with three hydrogens and two electrons remain as a lone pair.
Question – 5
5. Pick out the correct statement with respect to operative error
- A. A pipette calibrated at 200C cannot be used for 300C
- B. A person arrives at a wrong titre value because of confusion in the meniscus mark
- C. Use of improper indicator in volumetric analysis
- D. This error cannot be controlled by an experimentalist
- E. None of these
- Answer:B
- Answer Explanation:Operative errors are also called as personal errors and are introduced because of the variation of personal judgment. A person arrives at a wrong titre value because of confusion in volume of the meniscus mark. It is the personal error because for colored solutions, the titre value should be taken from the upper meniscus mark and for colorless solutions the titre value is from lower meniscus. The option A is an instrumental error and option C is a methodic error and option D is a random error.
Question – 6
6. Find out X in the following reaction.
- A. -CN
- B. -SO3H
- C. -NH3+
- D. -OCH3
- E. -COOCH3
- Answer:D
- Answer Explanation:The products of the reaction contain ortho and paraproducts. So, the X should be a molecule which can direct the incoming species to the ortho and parapositions. Generally, electron donating substituent on the ring directs the incoming group to ortho and parapositions. Here, the -OCH3 group is an electron donating group, which can direct the incoming electrophile to ortho and parapositions. The rest of the groups are an electron withdrawing in nature and direct the incoming electrophile to meta position.
Question – 7
7. Find out the emitted particle X from the reaction.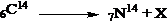
- A. ??-particle
- B. ??-particle
- C. ??-ray
- D. Positron
- E. Neutron
- Answer:B
- Answer Explanation:In the above reaction, the atomic number of the reactant is increased by one. It’s possible only by the emission of a beta particle. In beta particle emission, a neutron is changed to a proton and the atomic number of the element is increased. The net reaction in beta emission is as follows

Question – 8
I. It follows first order kineticsII. Polar aprotic solvent is a suitable solvent for the reaction.III. Product is obtained with the inversion of configuration to that of the reactant.
- A. I only
- B. II only
- C. III only
- D. I and III
- E. II and III
- Answer:E
- Answer Explanation:The rate of the SN2 reaction depends on the both the reactant and the nucleophile. So, it follows second order kinetics. Polar protic solvents slow down the reaction by the salvation process. But polar aprotic solvent doesn’t involve in the solvation process. In SN2 reaction the nucleophile attacks the reactant from the back side. Therefore, the structure of the product has the inversion of configuration that of the reactant.
Question – 9
9. Which of the following steps will have the highest ionization energy? 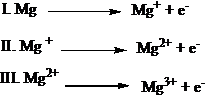
- A. I only
- B. II only
- C. III only
- D. Both II and III
- E. Both I and II
- Answer:C
- Answer Explanation:The step, in which Mg2+ is converted to Mg3+ will have the highest ionization energy, because Mg2+ has nobel gas configuration (1s2, 2s2, 2p6). Therefore, to remove an electron from the Mg2+ ion requires high energy.
Question – 10
10. How many signals will appear for the naphthalene molecule in 1H NMR spectrum?
- A. 8
- B. 6
- C. 4
- D. 3
- E. 2
- Answer:E
- Answer Explanation:Generally, in 1H NMR spectrum the number of signals equals to the number of different sets of protons present in the molecule. In a molecule, chemically equivalent protons give only a single signal in the spectrum. In naphthalene, there are eight hydrogens, in which four sets (??, ??) of chemically equivalent protons are available. Therefore, it will give two peaks in the spectrum.

Question – 11
11. The rate constant for a second-order reaction is 3.33 X 10-2 dm3 mol-1 s-1. If the initial concentration of the reactant is 0.05 mol dm-3, calculate its half life.
- A. 20 min
- B. 5 min
- C. 8 min
- D. 10 min
- E. 2 min
- Answer:D
- Answer Explanation:The half-life period for a second order reaction is, t1/2 = 1/(k2a)Given data:Rate constant k2 = 3.33 X 10-2 dm3 mol-1 s-1Initial concentration ‘a’ = 0.05 mol dm-3Therefore, t1/2 = 1/(3.33 X 10-2 dm3 mol-1 s-1 X 0.05 mol dm-3)= 1/(1.665 X 10-3 s-1)t1/2 = 600 s = 10 min
Question – 12
12. Calculate the volume of a solution in mL containing 0.05M of 2 g H2SO4.
- A. 40 mL
- B. 400 mL
- C. 4000 mL
- D. 40000 mL
- E. 4 mL
- Answer:D
- Answer Explanation:Concentration or strength of solution = (Mass of solute) / (Volume of solution)Given data:Mass = 2 g; strength = 0.05MVolume = Mass of solute / concentration of solution= 2 g / 0.05MVolume of the solution in litre= 40 LVolume of the solution in millilitre= 40 X 1000 = 40000 mL
Question – 13
13. Which of the following acids cannot be prepared by using Grignard reagent?
- A. Acetic acid
- B. Formic acid
- C. Propionic acid
- D. Butanoic acid
- E. Pentanoic acid
- Answer:B
- Answer Explanation:Formic acid cannot be prepared using Grignard reagent, because it contains only one carbon atom. The rest of the acids can be prepared using Grignard reagent since, they have more than one carbon atom.
Question – 14
14. How much heat is required to melt 500 g of ice?
- A. 1.17 X 105 J
- B. 1.77 X 10-5 J
- C. 1.54 X 105 J
- D. 1.22 X 10-5 J
- E. 1.67 X 105 J
- Answer:E
- Answer Explanation:The liquid water is formed by the energy of ice and heat of fusion of ice, thus involves the change of state.q = mass X heat of fusion of ice= 500 g X 333 J/gq = 1.67 X 105 J
Question – 15
15. Which of the following molecules does not exhibit geometrical isomerism?
- A. 1,2-ethylenedicarboxylic acid
- B. 2-pentene
- C. Diazines
- D. 1,1-dichloroethene
- E. None of these
- Answer:D
- Answer Explanation:Geometrical isomers are cis-trans isomers. All the molecules exhibit geometrical isomerism except 1,1-dichloroethene, because both the chlorine atoms are attached on the same carbon atom.