Question – 1
1. What is the measure of the bond angle in a benzene molecule?
- A. 450
- B. 600
- C. 900
- D. 1200
- E. 1800
- Answer:D
- Answer Explanation:In a benzene molecule, each of the six carbon atoms are sp2 hybridised. The sp2-hybridised carbon has a bond angle of 1200. Each of the carbons in benzene utilises two sp2 orbitals to form a bond with two other carbon atoms. The structure of benzene is planar.
Question – 2
2. Cis-trans isomers are considered to beI. EnantiomersII. DiastereomersIII. Stereoisomers
- A. I only
- B. III only
- C. Both I and II
- D. Both I and III
- E. Both II and III
- Answer:E
- Answer Explanation:Cis-trans isomers are considered to be diastereomers. Diastereomers are stereoisomers but they are not enantiomers. Diastereomersare isomers which do not possess mirror image relationship with each other. It may result from chirality or from cis-trans isomers; whereas, enantiomers exhibit mirror image relationship and they result from chirality only.
Question – 3
3. Which of the following is the strongest acid?
- A. CH3-CH2-CH2-CH2
- B. CH3-CH2-CH=CH2
- C. CH2=CH2-CH=CH2
- D. CH3-CH2-CH3
- E. CH???CH
- Answer:E
- Answer Explanation:Acetylene is the strongest acid followed by alkene and alkane. The carbon with sp hybridisation is the most electronegative compound followed by the carbons with sp2 hybridisation and sp3 hybridisation. The atom with greater electronegativity shows greater acidic character. It is called as hybridisation effect.
Question – 4
4. The process used for the separation of volatile substances from non-volatile substances is
- A. Distillation
- B. Evaporation
- C. Crystallisation
- D. Filtration
- E. Sublimation
- Answer:E
- Answer Explanation:Sublimation process is very useful for the separation of volatile substances from non-volatile substances. Sublimation is the process in which, when a solid substance is heated, it is directly converted to vapour state without melting.
Question – 5
5. When the reaction quotient ‘Q’ is greater than the equilibrium constant ‘Kc‘ under the same temperature and pressure, it means that the
- A. Forward reaction is favoured
- B. Reverse reaction is favoured
- C. Both a and b
- D. Forward reaction with catalyst is favoured
- E. None of these
- Answer:B
- Answer Explanation:When Q >Kc, reverse reaction is favoured. When Q < Kc forward reaction is favoured. These aspects will hold well only when Q and Kc are under the same conditions of temperature and pressure. The addition of catalyst has no effect on the state of equilibrium.
Question – 6
6. The actual volume consumed for standardization of KMnO4 is 23.46 mL. The values obtained by a student are 19.46 mL, 21.52 mL, 27.42 mL and 25.17 mL. Calculate its percent uncertainty.
- A. 15%
- B. 16%
- C. 17%
- D. 18%
- E. 19%
- Answer:C
- Answer Explanation:Uncertainty is a quantitative measurement to calculate how much of your measured values deviate from a standard or expected value.Percent uncertainty = (Range of values deviated from a standard value/actual value) X 100Range of values deviated from a standard value is calculated by actual value minus observed value; 23.46-19.46 = +4, 23.46-21.52 = +1.94, 23.46-27.42 = -3.96, 23.46-25.17 = -1.71. So, range of values deviated from a standard value lies within 4mL.Percent uncertainty = (4 / 23.46) X 100 = 17%
Question – 7
7. A gas expands against the external pressure of 5 atm from a volume of 8L to 13 L. How much work is done?
- A. +40 atm.L
- B. -65 atm.L
- C. +25 atm.L
- D. -25 atm.L
- E. -40 atm.L
- Answer:D
- Answer Explanation:Work W = P X ??V = 5 atm X (13L – 8L) = 25 atm.LThe work has been done by the system. Therefore, W = -P X ??V = -25 atm.L
Question – 8
8. What are the reagents used for the conversion of acetic acid to alkane?
- A. HI/P, ??
- B. H2/Ru, Pressure
- C. LiAlH4
- D. Oxidation
- E. Heating
- Answer:A
- Answer Explanation:By heating acetic acid in the presence of hydrogen iodide and phosphorus, it gets reduced to ethane by the elimination of two water molecules. The acid group gets reduced to primary alcohol, when it reacts with reducing agent like lithium aluminium hydride or H2/Ru under pressure. This reaction works under high pressure.
Question – 9
9. Choose the correct order of priority of the functional groups, on numbering a molecule.
- A. Acid > Ester > Nitrile > Aldehyde
- B. Ester > Acid > Nitrile > Aldehyde
- C. Acid > Ester > Aldehyde > Nitrile
- D. Ester > Acid > Aldehyde > Nitrile
- E. Aldehyde > Acid > Ester > Nitrile
- Answer:A
- Answer Explanation:On naming a compound with a multiple functional groups, the highest priority goes to the most oxidized functional group in the molecule. In the priority order, acid and acid derivatives get the highest ranks.
Question – 10
10. The pH of a solution is 8.2 at 250 C. Calculate it’s [OH–].
- A. 5.8 M
- B. 1.58 X 10-6 M
- C. 2.28 X 10-4 M
- D. 4.28
- E. 6.89 X 10-6 M
- Answer:B
- Answer Explanation:pH + pOH = 14.0 ; pOH = 14.0 – pHpOH = 14.0 – 8.2 = 5.8pOH = 5.8[OH]– = antilog [-5.8] = 1.58 X 10-6 M
Question – 11
11. The bond order for the molecule O2+ is
- A. 2
- B. 2.5
- C. 1.5
- D. 1
- E. 0
- Answer:B
- Answer Explanation:The atomic number of molecule O2+ is 15. The order of increasing energy of molecular orbitals is(??1s)2 (??*1s)2(??2s)2 (??*2s)2(??2pz)2 (??2px)2 (??2py)2 (??*2px)1.Bond order = (Nb – Na)/2 = (8-3)/2Bond order = 5/2 = 2.5
Question – 12
12. The change in the oxidation state of boron in the following reaction is
- A. 4
- B. 3
- C. 2
- D. 1
- E. 0
- Answer:E
- Answer Explanation:The oxidation state of boron in NaBO2 = (+1) + x + (-2 X 2) = 3The oxidation state of boron in Na2B4O7 = (+1 X 2) + 4x + (-2 X 7)= 3Therefore, the change in the oxidation of boron is zero
Question – 13
13. Which elements have the greatest tendency to lose electrons?
- A. Cesium
- B. Calcium
- C. Magnesium
- D. Radium
- E. Iodine
- Answer:A
- Answer Explanation:Cesium has the greatest tendency to lose its outermost 6s1 electron and attain a noble gas configuration of [Xe]. Calcium [4s2], magnesium [3s2] and radium [7s2] are stable elements because of completely filled s-orbitals. Iodine is also a stable element because of half filled 5p orbitals.
Question – 14
14. Which of the following compounds would react faster in SN2 reaction?
- A.
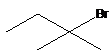
- B.

- C. CH3-CH2-CH2Br
- D. CH2=CH-CH2Br
- E.
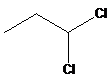
- Answer:D
- Answer Explanation:Generally, in SN2 reaction the transition state has a carbocation character and can be stabilised by resonance. The carbocation formed in allyl group is much more stable followed by primary, secondary and tertiary carbocations.
Question – 15
15. Complete the following nuclear reaction7N15 (p, ??)………
- A. 8O16
- B. 6C12
- C. 9F19
- D. 7N14
- E. 10Ne20
- Answer:B
- Answer Explanation:The nuclear reaction is to be written as follows,7N15 + 1H1 ??? 6C12 + 2He4